ML41176-BIO (AvL)
Unraveling tumor response and resistance to combined chemotherapy and PD-L1 inhibition with minimal invasive techniques in patients with advanced NSCLC with targetable disease
Enrollment
Recruiting
No. of patients
00 / 00
Population
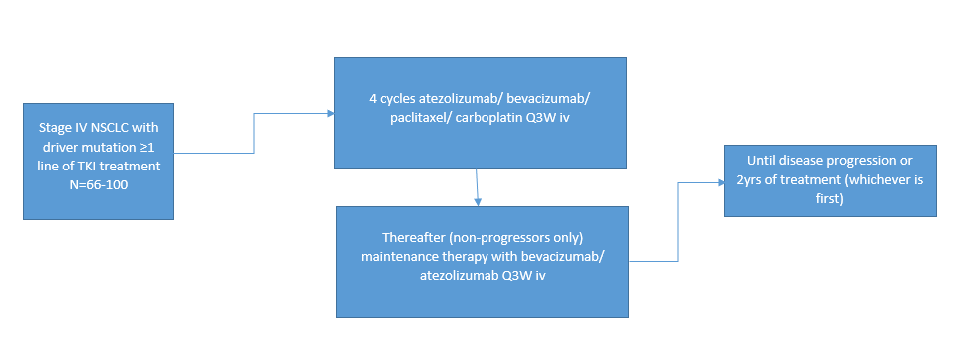
Stage 4 NSCLC patients with a tumor harboring a driver mutation and disease progression after at least one line of TKI treatment, >18 years old
Design
Single arm Explorative multicenter biomarker study, stratifying for resistance (primary resistance, acquired resistance, no resistance)
Key outcome parameters
Primary endpoints:
- Response to treatment defined as Progression Free Survival (PFS) in months according to RECIST v1.1 and divided into the following subgroups of response:
- Primary resistance (PFS < 6 mo)
- Acquired resistance (PFS between 6 and 24 mo)
- No resistance (PFS > 2 years)
Secondary endpoints:
- Overall survival
- Time to biological progression, defined as an increase of > 20% in the levels of the specific mutation from the lowest value achieved, measured in ctDNA in blood using ddPCR
- Immune related adverse events (irAE)
- Quality of life and symptom scores
Intervention
Standard chemo-immunotherapy (Atezolizumab/paciitaxel/ carboplatin/bevacizumab q3 wks iv)
Key inclusion criteria
Inclusion criteria
- Subjects with histologically- or cytologically-documented non squamous NSCLC with a documented driver mutation (such as EGFR, ALK, ROS, BRAF, MET, RET, NTRK1-3, KRAS, NRG1)
- Evidence of radiological disease progression following first line TKI or any subsequent treatment lines with TKI only (for EGFR treated with either osimertinib or failure of other TKIs 2nd line or later)
- Previous course of chemotherapy is allowed, but not necessary as well
- At least one lesion, not previously irradiated, that can be accurately measured at baseline as ≥ 10mm in the longest diameter (except for pathological lymph nodes they must be ≥ 15mm in short axis)
Key exclusion criteria
Exclusion criteria
- Prior treatment with CD137 agonists or immune checkpoint blockade therapies, anti−PD-1, and anti−PD-L1 therapeutic antibodies. Patients who have had prior anti−CTLA-4 treatment may be enrolled, provided the following requirements are met: Minimum of 6 weeks from the last dose of anti−CTLA-4. No history of severe immune related adverse effects from anti−CTLA-4 (CTCAE Grade 3 and 4)
- CNS disease, treated brain metastases without the need for steroids are allowed. Leptomeningeal disease
- Uncontrolled tumor-related pain
- Uncontrolled pleural effusion, pericardial effusion, or ascites requiring recurrent drainage procedures (once monthly or more frequently)
- History of autoimmune disease
- History of idiopathic pulmonary fibrosis, organizing pneumonia (e.g., bronchiolitis obliterans), drug-induced pneumonitis, idiopathic pneumonitis, or evidence of active pneumonitis on screening chest CT scan
- Significant cardiovascular disease, such as New York Heart Association cardiac disease (Class II or greater), myocardial infarction within the previous 3 months, unstable arrhythmias, or unstable angina
- Treatment with systemic immunostimulatory agents (including but not limited to IFNs, IL-2)
- Treatment with systemic immunosuppressive medications (including but not limited to prednisone ≥ 10mg, cyclophosphamide, azathioprine, methotrexate, thalidomide, and anti−tumor necrosis factor agents) within 2 weeks prior to inclusion of the study
- Inadequately controlled hypertension (defined as systolic blood pressure > 150 mmHg and/or diastolic blood pressure > 100 mmHg). Anti-hypertensive therapy to achieve these parameters is allowable
- Significant vascular disease (e.g., aortic aneurysm requiring surgical repair or recent peripheral arterial thrombosis) within 6 months prior to start of treatment
- History of hemoptysis (≥ one-half teaspoon of bright red blood per episode) within 1 month prior to start of treatment
- Serious, non-healing wound, active ulcer, or untreated bone fracture
Contact opnemen over een studie
Neem contact op voor meer informatie over de studies van de afdeling thoracale oncologie van Amsterdam UMC.