SIRP-alfa
An open label Phase I PET imaging study to investigate the bio- distribution and tumor uptake of [89Zr]Zr-BI 765063 in patients with head and neck squamous cell carcinoma, non-small cell lung cancer or melanoma who are also treated with ezabenlimab
Enrollment
Recruiting in Arm B only (BI 770371 i.p.v. BI 765063, en bijbehorende tracers)
No. of patients
4 / 22
Population
Patients with advanced, unresectable and/or metastatic NSCLC, HNSCC or melanoma, who exhausted standard therapies for the disease or who are unfit for standard therapies
Design
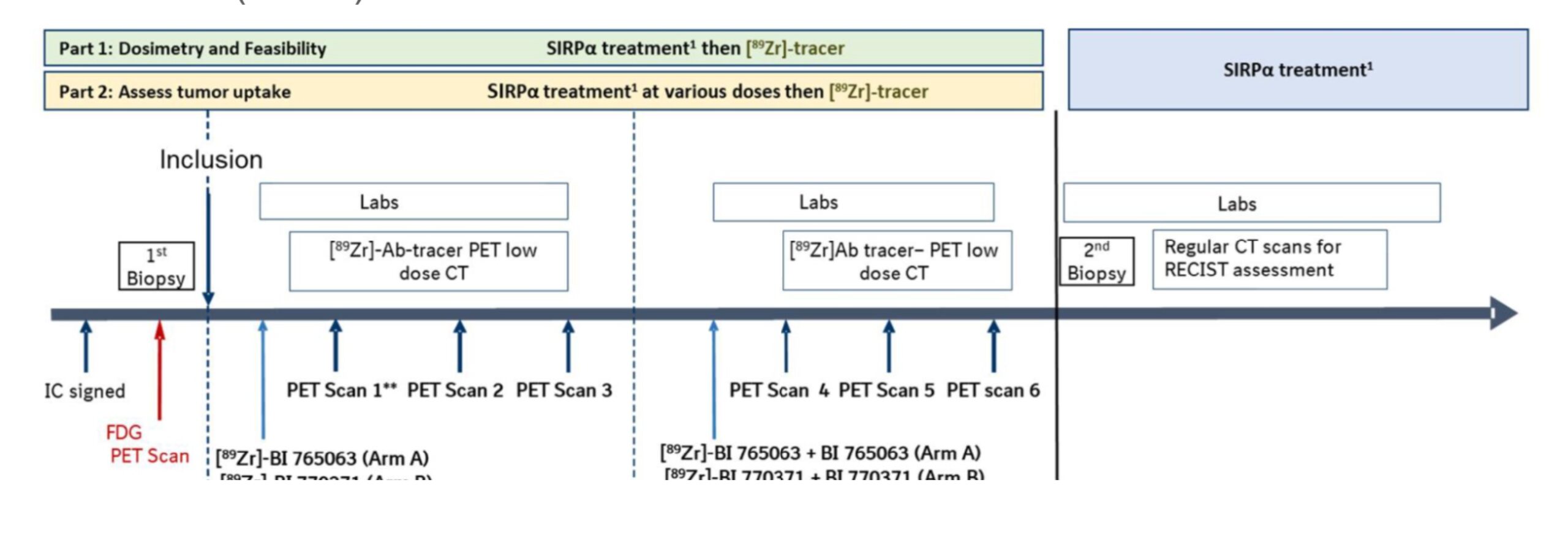
This is an open label, non-randomized bio-distribution trial, using a staggered approach:
- Part 1: dosimetry and feasibility assessment of [89Zr]Zr-BI 765063 (arm A) or [89Zr]Zr-BI 770371 (arm B) PET / CT.
- Part 2: assessment of tumor uptake of [89Zr]Zr-BI 765063 or [89Zr]Zr-BI 770371 (arm B) at various doses of BI 765063 or BI 770371) treatment
Key outcome parameters
Primary endpoint:
- Relative change in PET-uptake
Intervention
Each patient will undergo 2 phases of PET imaging assessment, followed by study treatment with BI 765063 (arm A) or BI 770371 (arm B) and BI 754091 (anti PD-1).
- Baseline assessment at Cycle 1
- On-treatment assessment at Cycle 2 + study treatment
Key inclusion criteria
Inclusion criteria:
- Male or female aged ≥ 18 years (no upper limit of age) at the time of ICF signature
- Eastern Cooperative Oncology Group (ECOG) performance status of 0 or 1
- Life expectancy of at least 3 months
- For arm A: SIRPα polymorphism V1/V1
- Patients with histologically or cytologically documented advanced/metastatic primary or recurrent HNSCC, melanoma, NSCLC who failed or are not eligible to standard therapy
- Patients with at least one measurable lesion are allowed as per RECIST v1.1
- Patient must have at least one PET imageable and evaluable tumor lesion with a diameter of at least 20 mm
- Patients must agree to on-treatment tumor biopsies (optional for first 3 patients in Part 1)
- Adequate biological parameters
- Prior major treatment-related surgery completed at least 28 days before study drug administration
- Interval of at least 28 days or 5 half-lives (whichever is shorter) since the last chemotherapy, approved immunotherapy, biological or investigational therapy, radiation or tyrosine kinase inhibitor (TKI) therapy must have elapsed before the first study drug administration(s) and all toxicities related to previous anticancer therapies have resolved to normal value or ≤ Grade 1 prior to the study treatment administration, except alopecia
- Capable of understanding and complying with protocol requirements
Key exclusion criteria
Exclusion criteria:
- Patients with symptomatic/active central nervous system (CNS) metastases;
- Other tumor location necessitating an urgent therapeutic intervention
- Presence of other active invasive cancers other than the one treated in this trial within 5 years prior to screening (or less, pending discussion with sponsor), except appropriately treated basal cell carcinoma of the skin, or in situ carcinoma of uterine cervix, or other local tumors considered cured by local treatment
- Patients with active autoimmune disease or a documented history of autoimmune disease, that requires systemic treatment (i.e. corticosteroids or immunosuppressive drugs); except patients with vitiligo, resolved childhood asthma/atopy, alopecia, or any chronic skin condition that does not require systemic therapy, patients with autoimmune- related hypothyroidism on a stable dose of thyroid replacement hormone and/or controlled Type 1 diabetes mellitus on a stable insulin regimen may be eligible
- Known severe infusion related reactions to monoclonal antibodies (Grade ≥ 3 National Cancer Institute (NCI)- Common Terminology Criteria for Adverse Events (CTCAE) v5.0) and patients removed from previous anti-PD-1 or anti-PD-L1 therapy because of a severe or life-threatening immune-related adverse event (irAE) (Grade ≥ 3 NCI-CTCAE v5.0)
- Patients receiving systemic treatment with any immunosuppressive medication within one-week prior to treatment start with BI 765063 and ezabenlimb; steroids of max. 10 mg prednisolone equivalent per day are allowed, topical and inhaled steroids are not considered as immunosuppressive
- Patients who have interstitial lung disease or active, non-infectious pneumonitis.
- Patients with uncontrolled disease-related metabolic disorders (e.g., hypercalcemia, SIADH) or uncontrolled diabetes
- Patients with uncontrolled congestive heart failure defined as New York Heart Association (NYHA) class III or IV, uncontrolled hypertension, unstable heart disease (e.g., coronary artery disease with unstable angina or myocardial infarction within 6 months before study treatment administration)
- Patients with significant chronic liver disease (e.g., significant fibrosis, known cirrhosis) or active hepatitis B virus (HBV) or hepatitis C virus (HCV) infection; if HbsAg positive, an effective antiviral treatment to prevent hepatitis B reactivation is recommended
- Patients with known Human Immunodeficiency Virus (HIV) infection or patients with an active infection requiring specific anti-infective therapy until all signs of infection have resolved, and this within 2 weeks prior to the first study treatment administration
- Women who are breastfeeding. Women who are breastfeeding can be enrolled if they stop breastfeeding. In this case, the patient will not be permitted to resume breastfeeding even after discontinuation of study treatment
- Patients whose medical, psychological including alcohol or drug abuse, or surgical conditions are unstable and may affect the study completion and/or compliance and/or the ability to give informed consent
Contact opnemen over een studie
Neem contact op voor meer informatie over de studies van de afdeling thoracale oncologie van Amsterdam UMC.